This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What is Amyotrophic Lateral Sclerosis (ALS)?
|
|
Amyotrophic lateral sclerosis (ALS) is a neurological disease that involves the degeneration and death of motor neurons necessary for voluntary muscle movement. Voluntary muscle movements can consist of chewing, walking, and other functions that are essential for daily activities. These motor neurons are key for communication between the brain and muscle fibers. ALS primarily affects both upper and lower motor neurons, in which both degenerate or die. In upper motor neurons, electrical impulses travel from the brain to the spinal cord. Lower motor neurons originate from the spinal cord and extend to skeletal muscles. As a result of the degeneration of these nerve cells, muscles begin to lose their function and deteriorate over time. Inevitably, the brain loses the ability to send these synapses and results in the loss of most voluntary movements [1].
|
Who is affected by ALS?
|
The Centers for Disease Control and Prevention estimated that roughly 14,000-15,000 Americans were living with ALS in 2016 [1]. ALS predominately affects males ages 55-75, but can appear in all races, demographics, and ages [1]. Many influential figures in the past century have been diagnosed with ALS. This list includes the New York Yankee Lou Gehrig and Professor Stephen Hawking who both eventually passed due to the condition. Lou Gehrig's diagnosis and sequential death have allowed many to refer to the condition as "Lou Gehrig's Disease".
|
Figure 1: Image showing New York Yankee first basemen and his famous influential quote.
|
What causes ALS?
Genetic Background
Familial ALS
For most cases, physicians are not able to understand the exact cause or factors associated with ALS. However, some cases do have a direct cause of the disease. In approximately 5-10% of cases, it is shown that ALS originates primarily through genetic heritage associated with various pathogenic variants in C9orf72, TDP-43, and FUS [2]. These cases are called Familial ALS and have well established pathological variants underlying the cause of the disease. However, the reason why neuron death occurs in these patients is relatively unknown.
Sporadic ALS
For the 90% of remaining cases, patients are diagnosed as having Sporadic ALS. Some individuals with ALS show various gene mutations that have originated de novo, which are spontaneous based changes caused by replication errors or potential environmental factors. These genetic aberrations can also lead to a malfunctioning human immune system, which may cause some cells to attack their own neurons leading to cell death [2]. One major factor is that protein mishandling may lead to an aggregate or buildup of abnormal forms of proteins that become trapped in the cytoplasm [2]. It is believed that this leads to a toxic protein that is playing some role in neuron cell death in both sporadic and familial cases. Researchers have also found that 40% of sporadic cases are characterized by elevated glutamate levels located in the spinal cord [3]. Chemical concentrations like higher levels of glutamate may be toxic to neural cells if a neuron is continuously exposed to them and environmental factors may be playing a role [2]. Scientists are still working on understanding the mechanism associated with high glutamate levels.
For most cases, physicians are not able to understand the exact cause or factors associated with ALS. However, some cases do have a direct cause of the disease. In approximately 5-10% of cases, it is shown that ALS originates primarily through genetic heritage associated with various pathogenic variants in C9orf72, TDP-43, and FUS [2]. These cases are called Familial ALS and have well established pathological variants underlying the cause of the disease. However, the reason why neuron death occurs in these patients is relatively unknown.
Sporadic ALS
For the 90% of remaining cases, patients are diagnosed as having Sporadic ALS. Some individuals with ALS show various gene mutations that have originated de novo, which are spontaneous based changes caused by replication errors or potential environmental factors. These genetic aberrations can also lead to a malfunctioning human immune system, which may cause some cells to attack their own neurons leading to cell death [2]. One major factor is that protein mishandling may lead to an aggregate or buildup of abnormal forms of proteins that become trapped in the cytoplasm [2]. It is believed that this leads to a toxic protein that is playing some role in neuron cell death in both sporadic and familial cases. Researchers have also found that 40% of sporadic cases are characterized by elevated glutamate levels located in the spinal cord [3]. Chemical concentrations like higher levels of glutamate may be toxic to neural cells if a neuron is continuously exposed to them and environmental factors may be playing a role [2]. Scientists are still working on understanding the mechanism associated with high glutamate levels.
Environmental Factors
There are various theories behind what causes these sporadic cases of ALS. These factors can include smoking, toxins, pesticides, viruses, and Beta-methylamino-Ialanine (BMAA). Recent epidemiological evidence has shown that areas of Guam and the Kii Peninsula of Honshu Island of Japan contained rates of incidence 50-100 times higher than other areas [4]. These higher rates are believed to be due to BMAA exposure. It was once believed that local Micronesian plants were the source of BMAA in these communities, but recent research has shown that cyanobacteria are the real source in these areas [4]. One study has also shown that BMAA levels are higher in affected individuals with ALS than healthy individuals [4]. This suggests that BMAA and other environmental factors can play some role in the neurodegeneration associated with ALS. These impacts may involve altering DNA structure or simply inhibiting structures associated with the neuron. It is important to note with these findings that mechanisms behind ALS are complex and for many cases no main direct cause can be determined.
What are the symptoms of ALS?
|
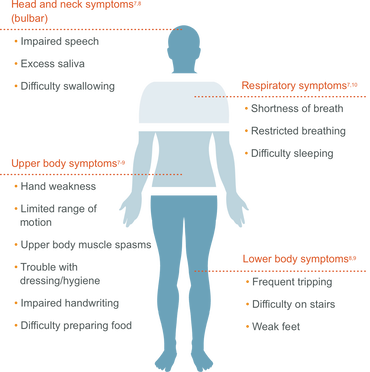
Figure 2: Illustration showing symptoms of ALS. Most cases begin with those symptoms associated with the upper extremity and soon develop many of the other symptoms listed in this figure.
|
Early signs of ALS include [2]:
ALS often starts in the upper extremity region primarily the hands, feet, or limbs [2]. Soon after, this weakness spreads to other areas of the body. After the nerve cells are destroyed, your muscles continue to weaken over time. This eventually begins to affect daily functions like chewing, swallowing, speaking, and eventually breathing. |
Are there any treatments for ALS?
|
There is no current cure for ALS. However, there are treatments that can help make symptoms manageable, prevent other medical complications from arising, and make daily activities easier as the disease progresses.
Current Treatment Options [1] :
|
Figure 3: ALS patient utilizing bars to perform a physical therapy exercise.
|
What is FUS?
|
FUS is a genetic region essential for providing the necessary instructions to make proteins and is found within the nucleus of many cell types [5]. Without FUS, many steps associated with the protein production would not be achieved. The FUS protein binds to a region of DNA associated with regulation of transcription [5]. In addition, this protein is important for processing molecules called messenger RNA or mRNA. These transcripts can be cut and rearranged in different ways, a process referred to as alternative splicing. Once this mRNA is processed, it is transported out of the nucleus to other cell structures that facilitate the creation of a mature protein. This protein has also been shown to help with various DNA repair mechanisms, which help prevent against the build up of DNA damage [5].
|
What role does FUS play in ALS?
|
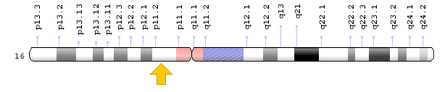
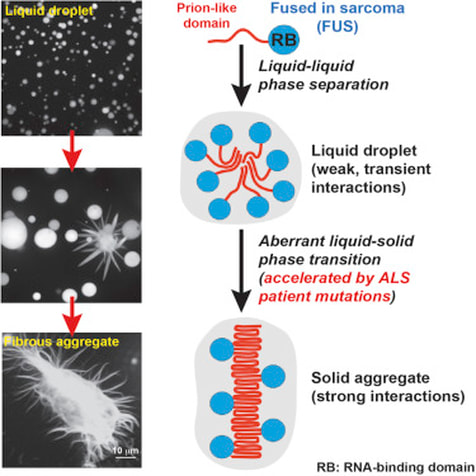
In 2009, a paper written by Kwiatkowski Jr. et. al suggested that 13 mutations in the FUS/TLS gene are associated with ALS [6]. So far as of today, at least 85 mutations have been found associated with the FUS gene and amyotrophic lateral sclerosis (ALS) [5]. Specifically, for FUS gene alterations, these cases tend to develop a more aggressive juvenile-onset form in comparison with individuals shown to have other gene alterations in C9orf72 or TDP-43 [5]. These genetic changes alter the DNA binding and mRNA processing ability of the protein. There has been evidence to suggest that the mutations interfere with the transportation of mRNA out of the nucleus in the cell [5,6]. Sharma et al. suggested that the severity of these mutations result from a new function that is detrimental to cell rather than the lack of proteins [7] Recent research has found that these aggregates are caused by a liquid-to-solid phase transition associated with the FUS protein [8]. The FUS protein can use a reversible liquid-to-solid transition, which plays a normal role in cell organization. However, mutations in the FUS gene create proteins that form fibrous aggregates when induced by stress. These proteins no longer can undergo this continuous transition and these stress granules may underlie the cause of ALS. It also suggested that DNA repair may play a role as many individuals show high amounts of DNA damage [6]. However, these results still fail to address their overall impact on this disease.
|
Figure 5: Visual depiction showing that the FUS protein has a prion-like domain that can undergo this liquid-to-solid phase contrast. Patient mutations lead to solid aggregates shown at the bottom of the figure. These become irreversible and suggest that this physical alteration plays a role in neurodegeneration [8].
|
Current Gaps in Knowledge:
Role of Protein Regulation/Degradation of FUS
Reason for FUS Related Neuron Death
Reason for Early-Onset with FUS
Role of FUS in DNA Repair
Long-Term Effects of Altered FUS-related DNA Repair Mechanisms
Reason for FUS Related Neuron Death
Reason for Early-Onset with FUS
Role of FUS in DNA Repair
Long-Term Effects of Altered FUS-related DNA Repair Mechanisms
ALS Non-Profit Organizations
Questions?
References
[1] Amyotrophic Lateral Sclerosis (ALS) Fact Sheet. (2018, August 9). Retrieved February 3, 2019 from https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Amyotrophic-Lateral-Sclerosis-ALS-Fact-Sheet
[2] Amyotrophic lateral sclerosis (ALS). (2018, July 17). Retrieved February 3, 2019 from https://www.mayoclinic.org/diseases-conditions/amyotrophic-lateral-sclerosis/symptoms-causes/syc-20354022
[3] Glutamate. (2019). Retrieved February 3, 2019 from http://web.alsa.org/site/PageServer?pagename=ALSA_Glutamate
[4] Ingre, C., Roos, P.M., Piehl, F., et al. (2015, February 12). Risk factors for amyotrophic lateral sclerosis. Retrieved February 3, 2019 from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4334292/
[5] FUS Gene-Genetics Home Reference. (2019, January 29). Retrieved February 3, 2019 from https://ghr.nlm.nih.gov/gene/FUS#
[6] Kwiatkowksi, T.J., et al. (2009, February 27). Mutations in the FUS/TLS Gene on Chromosome 16 Cause Familial Amyotrophic Lateral Sclerosis. Science, 323 (5918):1205-1208. DOI: 10.1126/science.1166066
[7] Sharma, A., Lyashckenko A.K., et al. (2016, February 4). ALS-associated mutant FUS induces selective motor neuron degeneration through toxic gain of function. Nature Communications, 7:104651. DOI: 10.1038/ncomms10465
[8] Patel, A., Lee, H.O., et al. (2015, August 27). A Liquid-to-Solid Phase Transition of the ALS Protein FUS Accelerated by Disease Mutation. Cell, 162(5): 1066-1077. DOI:https://doi.org/10.1016/j.cell.2015.07.047
Header Image: https://singularityhub.com/wp-content/uploads/2018/03/neurons-brain-1-1068x601.jpg
Figure 1: https://d2g8igdw686xgo.cloudfront.net/29846870_15261867800_r.jpeg
Figure 2: http://alspathways.com/wp-content/uploads/2017/08/chart-body-4.png
Figure 3: https://alstreatment.com/als-patient-josaine/
Figure 4: https://ghr.nlm.nih.gov/gene/FUS/location.png
Figure 5: https://www.cell.com/fulltext/S0092-8674(15)00963-0
[2] Amyotrophic lateral sclerosis (ALS). (2018, July 17). Retrieved February 3, 2019 from https://www.mayoclinic.org/diseases-conditions/amyotrophic-lateral-sclerosis/symptoms-causes/syc-20354022
[3] Glutamate. (2019). Retrieved February 3, 2019 from http://web.alsa.org/site/PageServer?pagename=ALSA_Glutamate
[4] Ingre, C., Roos, P.M., Piehl, F., et al. (2015, February 12). Risk factors for amyotrophic lateral sclerosis. Retrieved February 3, 2019 from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4334292/
[5] FUS Gene-Genetics Home Reference. (2019, January 29). Retrieved February 3, 2019 from https://ghr.nlm.nih.gov/gene/FUS#
[6] Kwiatkowksi, T.J., et al. (2009, February 27). Mutations in the FUS/TLS Gene on Chromosome 16 Cause Familial Amyotrophic Lateral Sclerosis. Science, 323 (5918):1205-1208. DOI: 10.1126/science.1166066
[7] Sharma, A., Lyashckenko A.K., et al. (2016, February 4). ALS-associated mutant FUS induces selective motor neuron degeneration through toxic gain of function. Nature Communications, 7:104651. DOI: 10.1038/ncomms10465
[8] Patel, A., Lee, H.O., et al. (2015, August 27). A Liquid-to-Solid Phase Transition of the ALS Protein FUS Accelerated by Disease Mutation. Cell, 162(5): 1066-1077. DOI:https://doi.org/10.1016/j.cell.2015.07.047
Header Image: https://singularityhub.com/wp-content/uploads/2018/03/neurons-brain-1-1068x601.jpg
Figure 1: https://d2g8igdw686xgo.cloudfront.net/29846870_15261867800_r.jpeg
Figure 2: http://alspathways.com/wp-content/uploads/2017/08/chart-body-4.png
Figure 3: https://alstreatment.com/als-patient-josaine/
Figure 4: https://ghr.nlm.nih.gov/gene/FUS/location.png
Figure 5: https://www.cell.com/fulltext/S0092-8674(15)00963-0
Recent Visits
Nathan Johnson
B.S. Genetics and Genomics
University of Wisconsin-Madison
[email protected]
Last Updated: 5/11/2019
B.S. Genetics and Genomics
University of Wisconsin-Madison
[email protected]
Last Updated: 5/11/2019